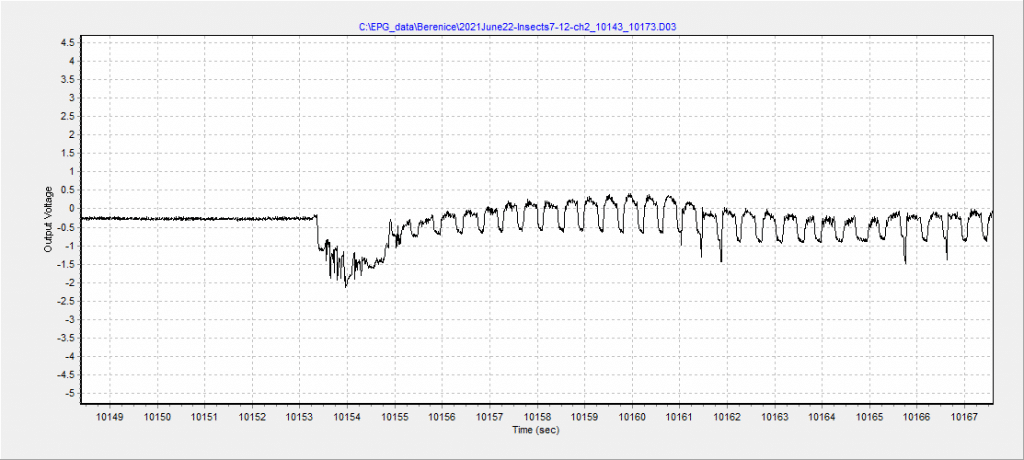
Key Result
This project is still in progress, but it aims to produce knowledge that will ultimately result in management recommendations for leafhoppers and aster yellows in multiple crop plants grown on the Canadian Prairies.
Project Summary
Background
Canola production can be affected by many insect pests and diseases, all of which can impact yield and cause economic losses to producers. This list of insects and diseases includes aster leafhoppers vectoring aster yellows (AY), which poses a major challenge to managing AY due to the migratory and enigmatic nature of the aster leafhopper vector. A recent characterization of different plant species commonly found in the Prairies indicated that canola is an unsuitable host for leafhopper development and reproduction. However, it’s unclear why canola is greatly affected by AY relative to other crops.
In plant diseases transmitted by vectors, feeding activity plays an important role in pathogen transmission and disease dynamics, as certain feeding events are responsible for acquiring or inoculating the pathogen into a host plant. This feeding activity has not been characterized for aster leafhoppers on crops grown on the Prairies. It’s possible that a shorter feeding time is sufficient for leafhoppers to transmit the AY pathogen to canola, while a longer time is required to transmit the pathogen to other crops. Additionally, plants can defend themselves against insect and/or pathogen attack, with plant hormones playing a large role in regulating defense responses.
With respect to AY and aster leafhoppers in canola, little is known about these plant hormonal responses or how they may regulate leafhopper attack. It is possible that canola is more affected by this disease because defense responses in this plant species are suppressed or weak compared to cereals. Understanding aspects related to leafhopper feeding, as well as if infection with AY changes these aspects, and how plant hormones mediate pathogen and leafhopper attack will provide valuable insights into AY epidemiology and help identify plant-defense traits that could be potentially incorporated in breeding programs. Furthermore, knowledge of the minimum times required to acquire or transmit aster yellows phytoplasma (AYp) can be directly converted into management recommendations, including the timing of insecticide applications.
Purpose
Both AY disease and leafhoppers can result in significant crop and yield loss, including field failure in extreme cases. Previous research indicates that aster leafhoppers do not reproduce on canola, yet this crop is severely affected by AY disease. One possible explanation is in the time required to transmit AY phytoplasmas (AYp), which is unknown for most plant species. Short periods of time could be enough for leafhoppers to transmit AYp to suboptimal plants such as canola and members of the Asteraceae family, while requiring a longer period for other plant species.
Quantification of the times required to acquire or transmit AYp can be directly converted into management recommendations. That is, it will allow the researchers to evaluate how much time between detection of insects may elapse before they will infect plants. Furthermore, if some trait such as activation or downregulation of a plant signaling pathway is found to be correlated with reduced insect oviposition, reduced feeding, impairment of insect development, and/or lower AY titer/milder symptoms, such knowledge could be incorporated into breeding programs to develop new crop varieties or enhance defense mechanisms in existing ones. This project will generate important information for the development of improved pest management practices for aster leafhoppers and AY disease, leading to reduced crop loss.
Objectives
This research has two objectives:
- To quantify the feeding behavior of aster leafhoppers on different host plants and examine the relationship between feeding time and AYp titer using the Electrical Penetration Graph technology followed by molecular quantification of phytoplasma titer via, LAMP, qPCR and ddPCR of plant tissue to quantify AYp titer.
- To establish feeding and reproductive preferences of aster leafhoppers using Arabidopsis thaliana lines in which their defense mechanisms are activated or silenced.
For more information on this topic, see Investigating the role of plant hosts in the outbreaks of the aster leafhopper vectored aster yellows and these other Aster yellows research projects on the Canola Research Hub.